By A Mystery Man Writer

I'm taking spectroscopy and instrumentation. Can someone tell me why the shorter the lifetime an atom is in an excited state, the wider the peak is on the spectra? I feel like

What is the maximum number of emission lines when the excited electron of an H atom in n =6 drops to the ground state?

The figure below represents part of the emission spectrum for a one-electron ion in the gas phase. All the lines result from electronic transitions from excited states to the n = 3

Hydrogen atom in ground state is excited by a monochromatic radiation of `lambda = 975 �`.

Excited state spectroscopy (kT ≈ 3.4 µeV), showing that the feedback
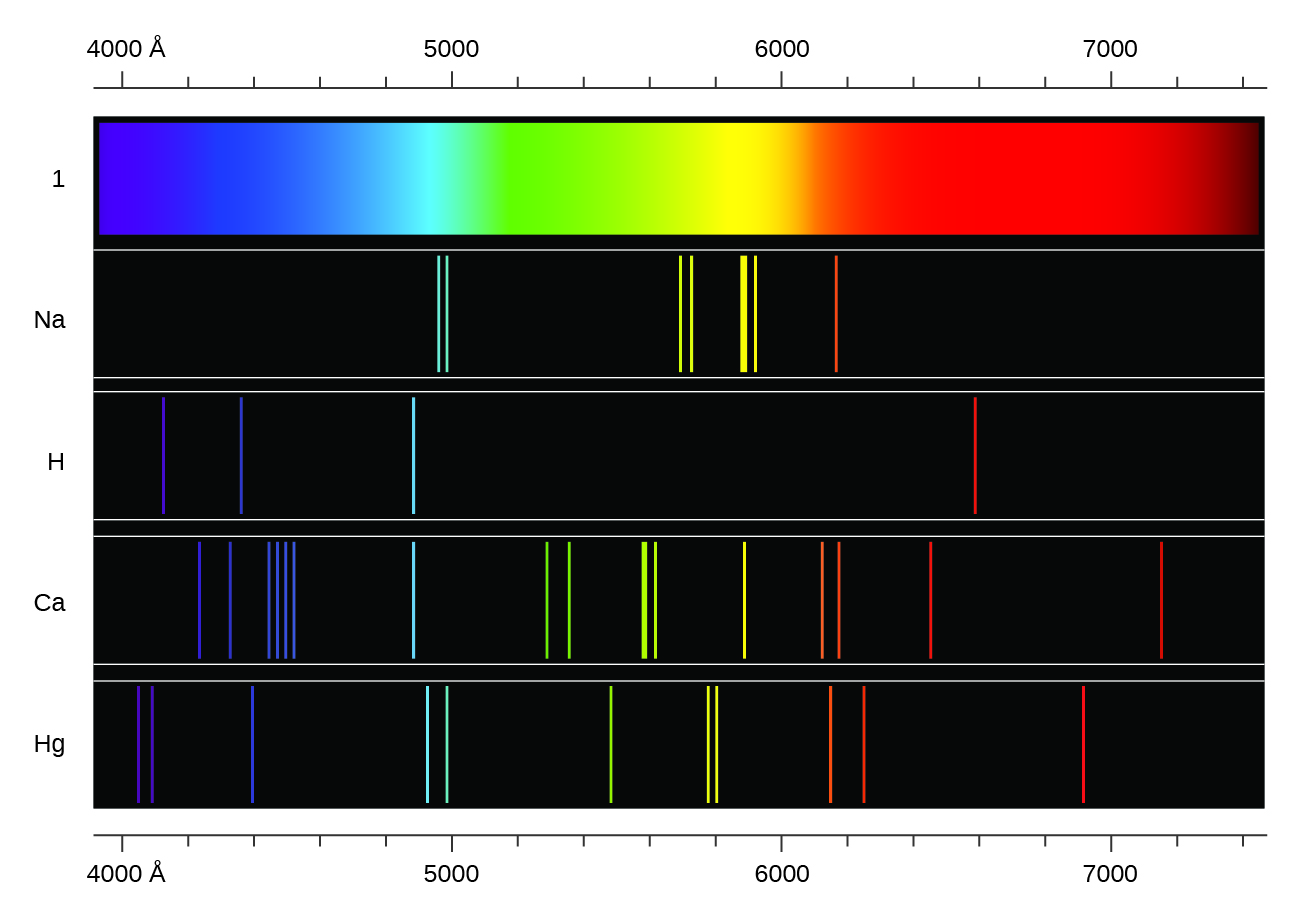
Hydrogen Emission Spectrum, Bohr Model of Hydrogen
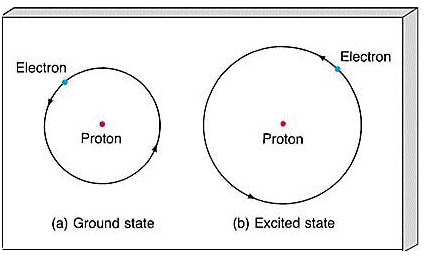
Chapter 4, Section 2
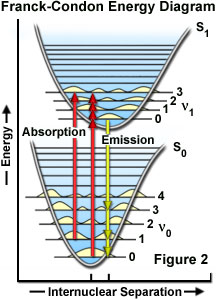
Confocal Microscopy - Fluorescence Excitation and Emission Fundamentals
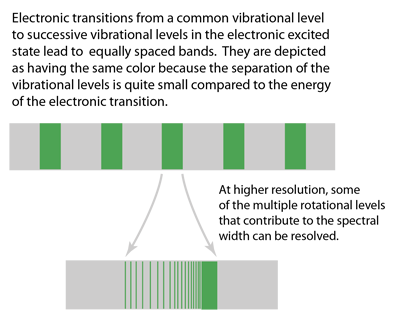
Electronic Spectra of Molecules

Lesson Explainer: Emission and Absorption Spectra

Solution] Bohr model: Absorption/Emission Spectra
A sample contains only hydrogen atoms and in all atom electron are present at 4th excited state. How many minimum number of atoms are required if total 7 lines are observed in
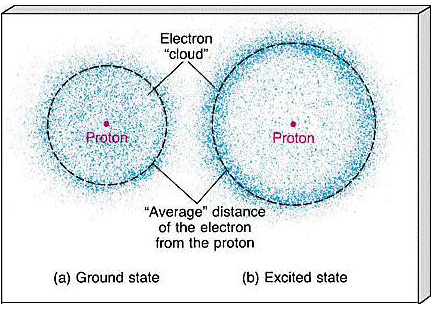
Chapter 4, Section 2

Absorption And Emission Line Spectra - Electronic Structure - MCAT Content

Chapter 4, Section 2